Unprecedented
Is Wes right about the Kent meningitis outbreak?
Wes Streeting has expressed concern about the rapid spread and severity of the meningitis outbreak in Kent. According to UKHSA health officials, the number of confirmed cases has risen from 15 to 20. Streeting described the outbreak in Kent as unprecedented. Therefore, we thought we’d take a look at the data.
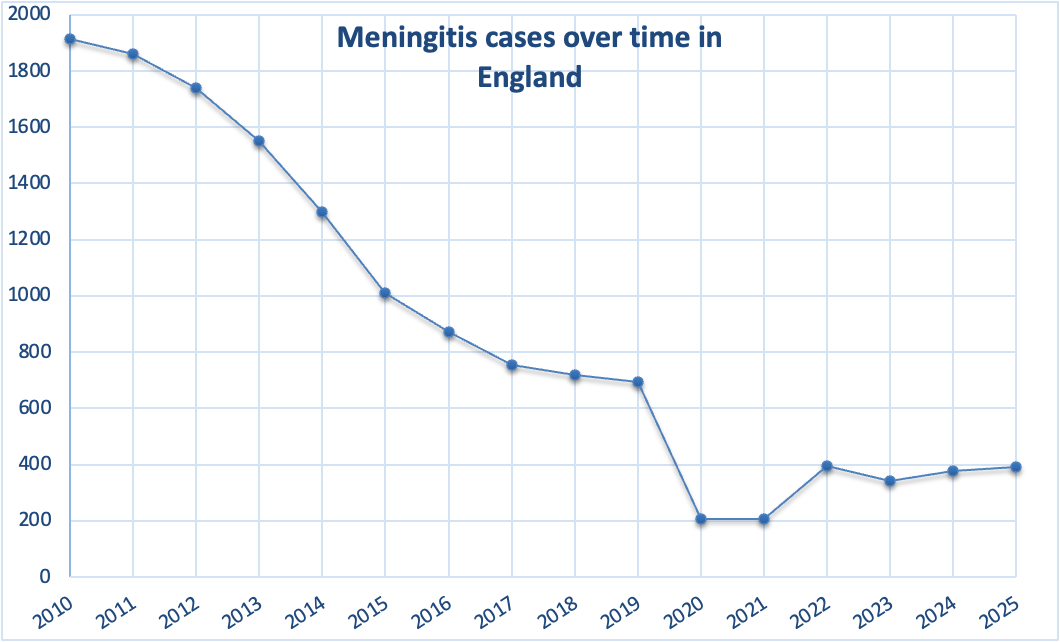
Based on UKHSA annual surveillance reports (2010–2025), the number of meningococcal cases in England decreased significantly, from approximately 1,915 in 2010 to 1,010 in 2015. This downward trend continued more gradually, reaching below 700 cases by 2019. In 2020, the number of cases dropped dramatically, largely due to COVID-19 restrictions. Following this decline, cases began to rise again, stabilising at roughly 340-400 per year.
Epidemiological years (July–June) were standardised and labelled by the starting year for consistency.
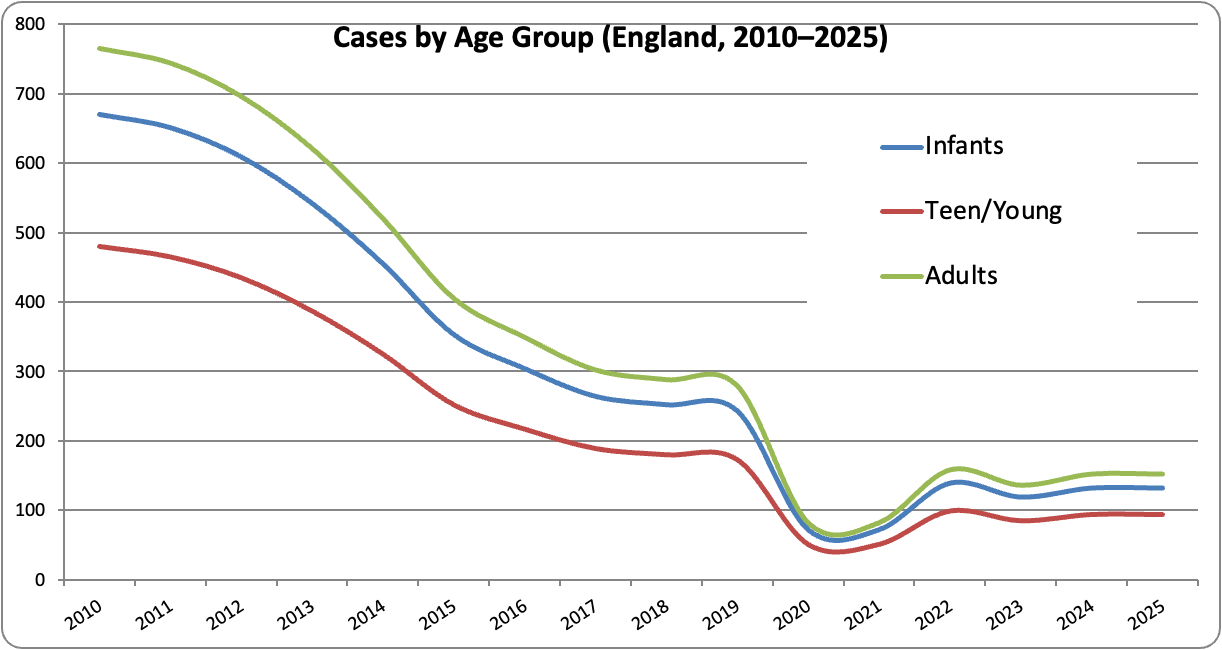
The case numbers for infants declined markedly from 2010 to 2019, dropped sharply in 2020, and then rose again post-pandemic. Teenagers and young adults formed the second-largest group, following a similar pattern of decline. Adult cases were lower overall but still showed the same temporal trend.
Across all age groups, cases decreased substantially over the decade, with a clear disruption in 2020 and partial recovery thereafter.
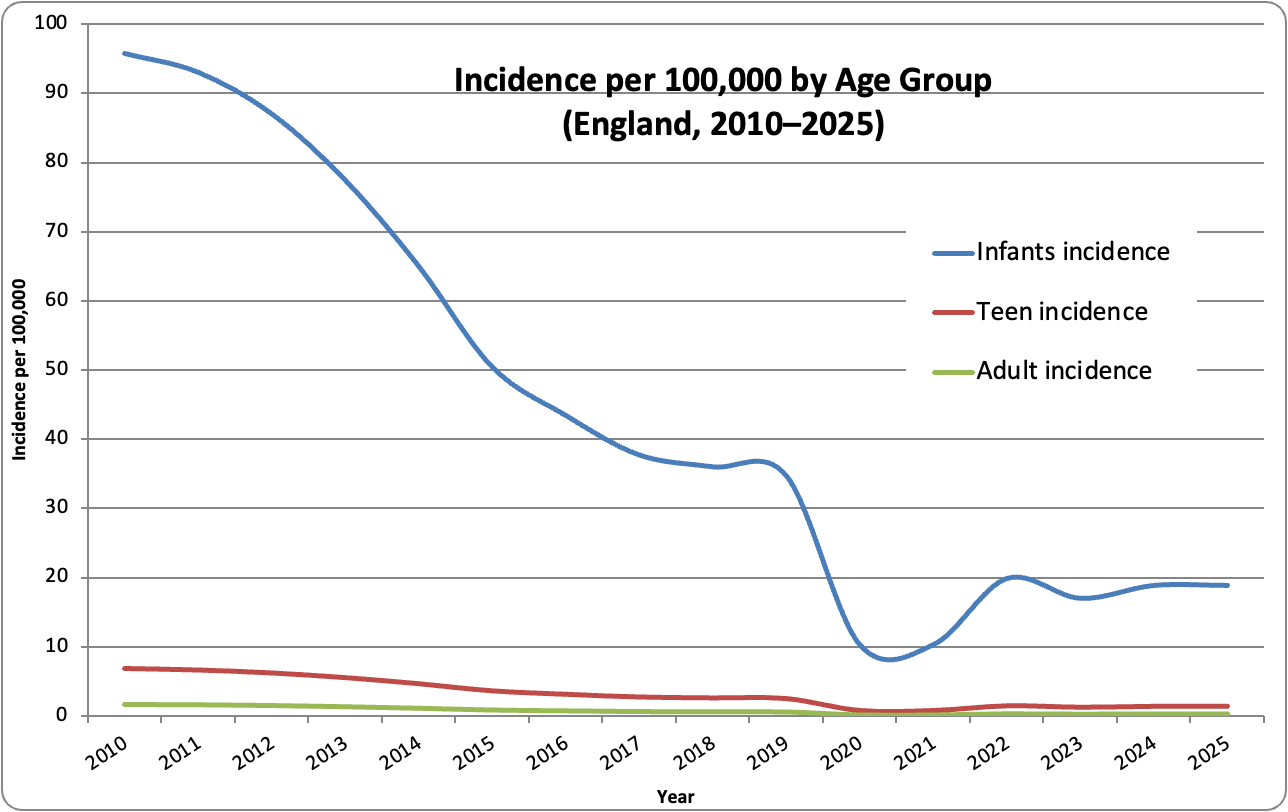
Making some assumptions about the age distribution of the population, infants have a roughly 15–60 times higher incidence than adults, who have a very low baseline risk.
Although overall UK meningitis incidence has fallen since 2010, there have been several notable outbreaks and clusters, mostly localised rather than nationwide epidemics.
Between 2015 and 2019, several UK universities, including Nottingham, Bristol and Edinburgh, reported localised clusters of meningococcal disease among students. These outbreaks were typically small but intense, often involving serogroups B or W, and were linked to close-contact environments such as halls of residence.
In 2015, students were urged to get vaccinated after a spike in meningitis cases. Figures reported 209 cases of MenW in 2015, with 22 linked deaths. The explicit warnings were targeted at students heading to university, who were considered a high-risk group. Liz Brown, chief executive of the charity Meningitis, said: “In the UK, every university could experience at least one case of meningitis amongst its students within the first term”.
Since COVID-19, the UK has not experienced large-scale meningitis outbreaks but has seen recurrent localised clusters, particularly among young people.
For example, a cluster of six cases was reported in the South of England in 2023, prompting targeted vaccination advice for students.
National surveillance data also show a rise in cases following the lifting of pandemic restrictions, reflecting renewed transmission rather than a single epidemic event. Public health warnings also highlighted increased risk among university students due to close-contact living environments. Together, this evidence indicates that meningitis remains endemic in the UK, characterised by repeated small clusters rather than widespread headline outbreaks.
The Kent 2026 meningitis outbreak differs from previous UK patterns in both scale and dynamics.
Earlier incidents were typically small, localised clusters—often involving 5–10 cases over several weeks—and were managed through routine public health responses such as contact tracing and prophylactic antibiotics.
In contrast, the Kent outbreak involved around 20 cases within a very short timeframe and was linked to a single social network. At least 10 cases attended Club Chemistry in Canterbury on 5, 6 or 7 March. It also resulted in multiple severe cases and 2 deaths, prompting escalation to a major public health incident. This contrasts with the usual endemic pattern of slower, contained transmission seen in the UK since 2010.
So, in answer to the question, the Kent outbreak is not unprecedented in the context of UK meningococcal disease, as localised clusters have occurred previously. However, it is unusual due to its rapid onset, relatively large number of cases within a single social network, and severity, distinguishing it from the typically smaller and slower-moving clusters observed in previous years.
This post was written by two old geezers who thought one of the functions of the UK Civil Service was to inform politicians before they made statements.
Methods
Data on invasive meningococcal disease (IMD) cases in England from 2010 to 2025 were compiled from UKHSA annual laboratory-confirmed reports and earlier Public Health England (PHE) surveillance publications. These sources provide national-level confirmed case counts based on routine surveillance systems. Additional context on long-term trends was supported by published epidemiological analyses (Subbarao et al., 2021).
Age-specific case data were standardised into three categories: infants (<1 year), teenagers and young adults (15–24 years), and other adults (25+ years). Where consistent age-stratified data were not available across all years, distributions were reconstructed using established epidemiological patterns reported by UKHSA. Incidence rates per 100,000 population were calculated using fixed population estimates for each age group to enable comparison over time.
This approach assumes relatively stable population sizes and age distributions across the study period. Furthermore, some data, particularly for earlier years, were derived from reconstructed proportions due to inconsistencies in historical reporting formats. As a result, the dataset may contain minor inaccuracies; however, these are unlikely to substantially affect the overall interpretation of temporal trends.
References
UK Health Security Agency (UKHSA) (2021–2025) Invasive meningococcal disease in England: annual laboratory-confirmed reports. Available at: https://www.gov.uk/government/collections/meningococcal-disease-guidance-data-and-analysis (Accessed: 18 March 2026).
Public Health England (PHE) (2021). Invasive meningococcal disease in England: annual laboratory confirmed report for epidemiological year 2019 to 2020. London: PHE. Available at: https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/951142/hpr0121_imd-ann.pdf (Accessed: 18 March 2026).
Subbarao, S. et al. (2021) ‘Invasive meningococcal disease, 2011–2020, and impact of the COVID-19 pandemic, England’, Emerging Infectious Diseases, 27(9), pp. 2495–2497.



Instantly brings to my mind at least the good old riddle of transmission of "disease".
Precisely why and how did some sadly die and others become ill in varying degrees.
Where did it ie the bacterial meningitis come from. Why are most unaffected.
This line of thought is absolutely crucial to understanding the riddle.
But it has never been done.
I wonder why not.
Considering Nick Rendell's comment below, I'll add that meningitis can be a rare adverse event following various vaccines. There are 2,126 VAERS reports of meningitis following various vaccinations: https://www.medalerts.org/vaersdb/findfield.php?TABLE=ON&GROUP1=AGE&EVENTS=ON&FOLLOWOPTIONS=LATEST&SYMPTOMS=Meningitis+%2810027199%29
There are several issues with the reporting system.
1) Obviously, we have no idea which of these (or any VAERS reports) were coincidences, or fully caused by the vaccine, or were situations where the vaccine played a triggering role along with a pre-existing condition.
2) A basic search on meningitis doesn't sort out which vaccine[s] was [were] reported with which event.
We would have to do a separate search for each vaccine, using the specific search terms for diagnosis and diagnostic codes to find that out.
That's a problem, because:
3) There are at least 22 different ways to report and code meningitis as a potential vaccine reaction:
Meningitis (10027197)
Meningitis aseptic (10027201)
Meningitis bacterial (10027202)
Meningitis borrelia (10049078)
Meningitis chemical (10027206)
Meningitis coxsackie viral (10027208)
Meningitis cryptococcal (10027209)
Meningitis echo viral (10027231)
Meningitis enteroviral (10027233)
Meningitis Escherichia (10084029)
Meningitis haemophilus (10027241)
Meningitis herpes (10027242)
Meningitis meningococcal (10027249)
Meningitis mumps (10027250)
Meningitis neonatal (10058780)
Meningitis noninfective (10058760)
Meningitis pneumococcal (10027253)
Meningitis salmonella (10027254)
Meningitis staphylococcal (10027255)
Meningitis streptococcal (10027256)
Meningitis tuberculous (10027259)
Meningitis viral (10027260)
There are also codes for
meningism (10027197)
meningeal disorder (10061281)
meningeal neoplasm (10061282)
meningeal thickening (10083667)
meningococcal bacteraemia (10058858)
meningococcal carditis (10027270)
meningococcal infection (10027274)
meningococcal sepsis (10027280)
meningoencephalitis bacterial (10051256)
meningoencephalitis herpetic (10027285)
meningoencephalitis viral (10074672)
meningoencephalitis herpes (10074249)
Notice the 2 separate codes for "meningoencephalitis herpetic" and "meningoencephalitis herpes," which appear to be identical conditions.
I can't quite make up my mind as to whether whoever designed this reporting system was spectacularly incompetent or whether they wanted to make sure nobody could find relevant reports (assuming the incident was reported, which is, sadly, rare even when the incident was likely strongly associated with the vaccine[s].
Either way, the result is the same. The data we need is to know whether something is or is not going on is not there.