Exploring regulatory data sets of the Comirnaty vaccine - 12a
What does the FDA package insert and SmPC report about the harms outcomes that matter?
The Package inserts provide warnings and precautions that report those serious side effects that might occur, and the recipient of the vaccine should be made aware of them.
The European regulators consider myocarditis and pericarditis “very rare” (< 1/10 000), whereas the FDA report the chance is “very low”. The FDA considers serious, low-frequency adverse events generally to be listed when there is reason to suspect that the drug may have caused the event. Both sets of regulators report that the risk is highest in young males: the FDA in males aged 12 through 17, whereas EMA and the MHRA report the risk up to 29 years old. Limited data indicates the risk in younger children is lower than in older children.
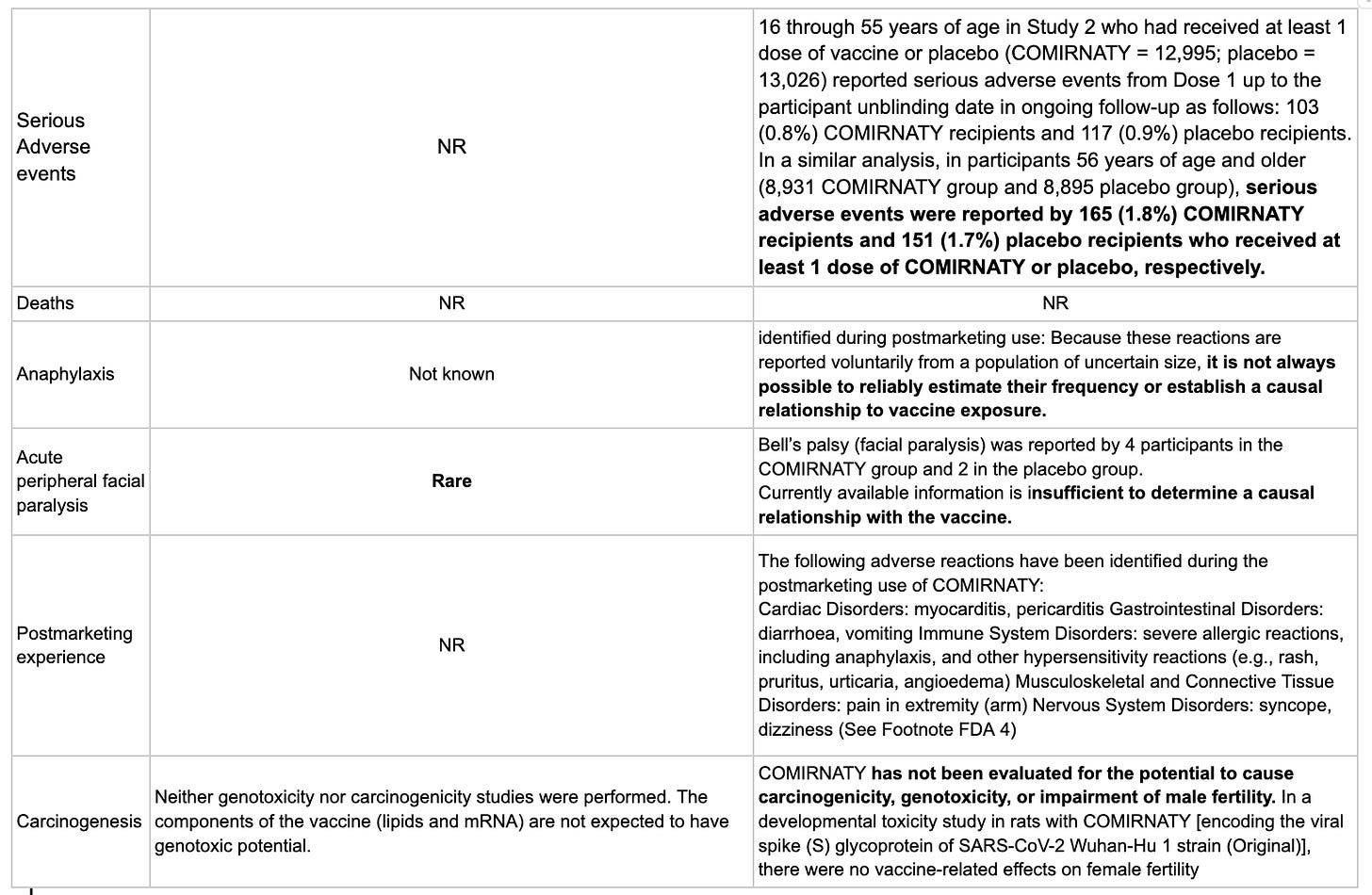
There is no information on death, and as we previously reported in post 11b, it is wrapped up in a composite outcome, which means there is no interpretable data. We also found no information on hospitalisation or secondary complications beyond the FDA report that “serious adverse events were reported by 165 (1.8%) COMIRNATY recipients and 151 (1.7%) placebo recipients who received at least 1 dose of COMIRNATY or placebo, respectively.”
European regulators consider facial palsy rare (≥ 1/10 000 to < 1/1 000). “The FDA report Bell’s palsy (facial paralysis) was reported by 4 participants in the COMIRNATY group and 2 in the placebo group. Currently available information is insufficient to determine a causal relationship with the vaccine.”
A host of adverse events were reported by the FDA in the post-marketing phase, including Cardiac Disorders: myocarditis, pericarditis Gastrointestinal Disorders: diarrhoea, vomiting Immune System Disorders: severe allergic reactions, including anaphylaxis, and other hypersensitivity reactions (e.g., rash, pruritus, urticaria, angioedema) Musculoskeletal and Connective Tissue Disorders: pain in extremity (arm) Nervous System Disorders: syncope, dizziness. There were no postmarketing results reported by the MHRA or EMA.
The FDA reports that COMIRNATY has not been evaluated for the potential to cause carcinogenicity, genotoxicity, or impairment of male fertility. European regulators report that “neither genotoxicity nor carcinogenicity studies were performed. The components of the vaccine (lipids and mRNA) are not expected to have genotoxic potential.”
We are still puzzled by all this. Why? Because events such as insomnia and anorexia (both possible effects on the central nervous system appear in the MHRA but not in the other sources we consulted (see the spreadsheet). The estimated incidences of many events are also not directly comparable, as we have already pointed out:
To sum up, package inserts are not that useful. There are worrying discrepancies overall, which is strange considering that the regulators and the NHS should all be looking at the same data. The package inserts reveal that postmarketing surveillance is non-existent. The issue of myocarditis in young people and children is concerning and should have warranted warnings. Overall, the regulator's approach to safety and informing the public is derisory.
We have previously written that reporting Adverse Drug Reactions should come with a Warning. Duplication of efforts leads to inconsistencies in patient information and fosters confusion. The costs of a flawed regulatory system are substantial, and failing to act in the best interests of patients will result in further unnecessary harm.
This post was written by two confused old geezers who wonder if regulators and other authorities are in danger of passing on to consumers the same information from the same data set.
Postscript: Adverse reactions observed during clinical studies are listed by the MHRA and EMA according to the following frequency categories: Very common (≥ 1/10), Common (≥ 1/100 to < 1/10), Uncommon (≥ 1/1 000 to < 1/100), Rare (≥ 1/10 000 to < 1/1 000), Very rare (< 1/10 000), Not known (cannot be estimated from the available data).







I am totally mystified why intelligent, esteemed doctors and scientists are losing their jobs or have their reputations smeared. What the hell is going on? I’ve started to make a list of the ones I’m aware of. Even two family court judges here in Ontario who ruled against the parent who wanted their children vaccinated, got their reputations smeared in the Toronto Star and Disciplined by the appeals court all because they determined there wasn’t enough evidence in front of them to rule in favour of the parent who wanted their children to get the shot. Statistician Norman Fenton in the U.K who has seen how the stats have been calculated incorrectly has also been the victim of ruthless career bashing.
In the article posted here “the Lipid nano particles in the mRNA shots are not expected to have genotoxic potential.” Are these people who make these sweeping conclusions inept? Captured by big pharma? Go to the manufacturer for answers? Or maybe all apply. Kevin McKernan who was the lead R&D on the human genome project and has a substack under the name Anandamide explains all he has found but it’s way over my head. He was the first to detect contaminated plasmid DNA, SV40. Dr. Phillip Buckhaults molecular biologist and cancer geneticist at South Carolina University didn’t believe it and tested it himself. Sure enough, he too found it and presented at a SC senate hearing on his discoveries. He has his IRB and is currently testing tissue samples for genome integration. Other discoveries include the double stranded RNA, N1 Methyl pseudouridine. People at the top of their game are screaming from the rafters to remove these shots. I don’t understand why there isn’t even a pause. Instead, let’s give them to babies. Something is seriously wrong.
A close friend and former colleague joined up to give the COVID vaccinations. She became very uncomfortable with the disregard to people's adverse reactions to the previous dose .When checking with person in charge was told to give the next dose. Like me she had been involved in childhood vaccination where caution was applied if reactions more severe than redness at site.